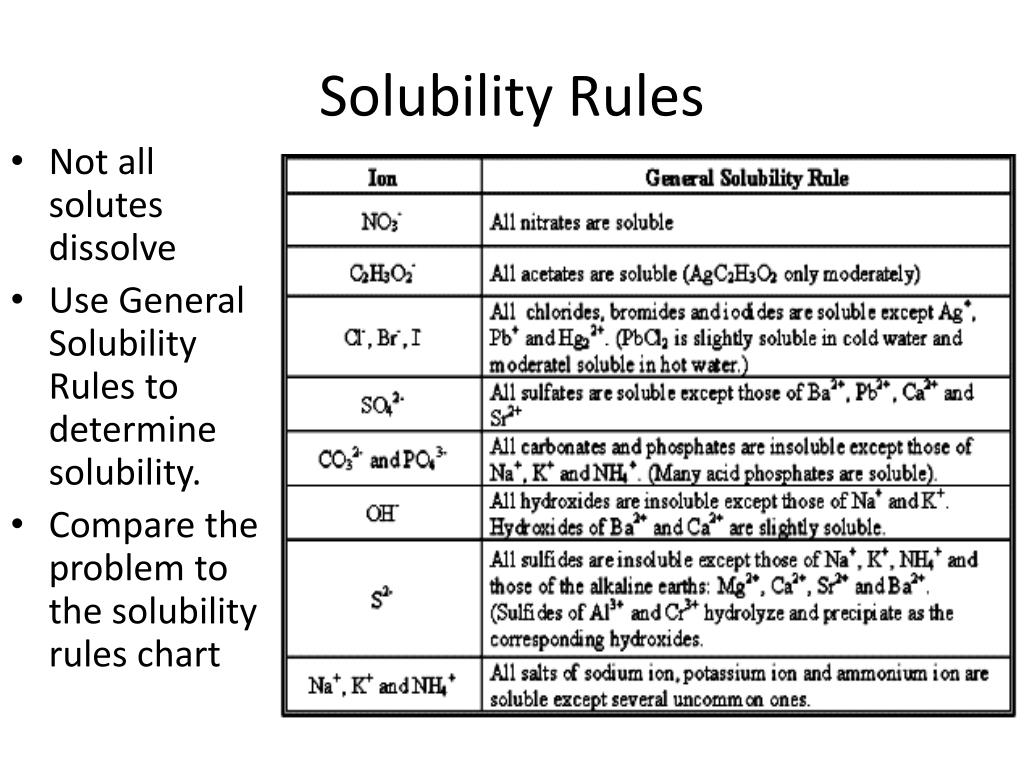
We describe an activity-based approach for teaching aqueous solubility to introductory chemistry students that provides a more balanced presentation of the roles of energy and entropy in dissolution than is found in most general chemistry textbooks. The precipitation reactions are directly the linkage with gravimetric analysis and Mohr, Fajans and Volhard methods in this context of volumetric analysis as well. It can be said that the models are hypothetically suitable to investigate the relations between the qualitative analysis and the quantitative analysis with the chemical calculation in this context the reactions of first group cations with HCl that is the group reagent of them. The solutions of problems have been done by two different methods that are known as by the using formula and by the conversion factor. Third Model: The aim of Third Model is to teach how to calculate the mass of precipitate if one of two reactants reacts completely and other remains unreacted when the molarities and the volumes of their solutions are given in the problems. Second Model: The aim of Second Model is to teach how to calculate the mass of precipitate if two reactants react completely when the molarities and the volumes of the solution of them are given in the problems. First Model: The aim of First Model is to teach how to calculate the mass of precipitate when the molarity and the volume of the solution of only one reactant is given in the problems. These sub models are consecutively called First Model, Second Model and Third Model. On these precipitation reactions, it is offered the three sub model for teaching how to calculate the quantity of precipitate if the quantities of one or two reactants are given for the quantitative analysis. For this aim, it is preferred the precipitation reactions of first group cations (Ag 1+, Pb 2+, 2 2 Hg) with HCl solution that is the group reagent in the qualitative analysis of inorganic samples in this study. The aim of this study is hypothetically to model the linkage between the qualitative analysis and the quantitative analysis with the chemical calculations. On the science or the chemistry education, it is one from the most important problems for both the science teachers and the students to design the linkage between ones information and other information as well. Application of DES as novel co-solvent with other organic solvents can be translated to economic benefits through reducing the energy consumption and operational risks associated with the use of flammable solvent such as n-hexane. On the other hand, the addition of DES to n-hexane can slightly improve the extraction yield and significantly reduce the extraction temperature in comparison to n-hexane alone. The addition of DES to THF negatively impacted oil yield. Different organic solvents were screened and it was found that n-hexane and THF can achieve the highest oil yield. It was noted that the solvent share the same non-polar region on the σ-profiles with the flaxseed oil model. COSMO-RS, UFF and DFT computational method was used to study the solubility of organic solvents and DESs for oil extraction from flaxseed. Enhancement of solvent extraction was investigated by adding novel co-solvents in the form of DESs to tetrahydrofuran (THF) and n-hexane. The extraction of oil from flaxseeds were carried out using deep eutectic solvents (DESs) and organic solvents with ultrasound assisted extraction. Keywords (Audience): High School / Introductory Chemistry A single rule for solubility is presented that is almost as accurate as the sets of rules presented in most textbooks. (iii) Solubility should be explained in terms of simple physical principles. (ii) If one desires to classify salts as soluble or insoluble in water, the criteria for this classification should be made clear. Examples of real solubility data should be given such that readers understand that degrees of solubility are possible. In addition, it is proposed that the following changes be made to the discussion of solubility so that students and teachers can have a greater understanding of solubility: (i) Solubility should not be presented in a strictly dualistic manner of soluble versus insoluble. 
Many general chemistry textbooks have errors or omissions in their rules for predicting solubility that the author highlights and attempts to correct. This article examines the precipitation and solubility of ionic compounds in water from seven general chemistry textbooks and the CRC Handbook of Chemistry and Physics.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
